
Can also used in molecular and atomic physics, astronomical spectroscopy and remote sensing.Ītomic absorption spectra and molecular absorption spectra. These dark bands are used to determine the absorption of the object.Įmission is the ability of a substance to give off light, when it interacts with heat.Ībsorption is the opposite of emission, where energy, light or radiation is absorbed by the electrons of a particular matter.Ĭan be used as part of spectroscopy to figure out the composition of a certain matter.Ĭan be used as part of spectroscopy to figure out the absorption level of certain objects and their ability to retain heat. The absorption spectrum is usually light colored, with dark bands that run through it. In order to determine the absorption level of a substance, a beam of radiation is directed at the sample and the absence of light that is reflected through the object can be used to calculate the absorption. They can also depend on temperature, electromagnetic field, interaction between the molecules of the sample, crystal structure in solids and temperature. Absorption is primarily determined by the atomic and molecular composition of the material. They are also used in molecular and atomic physics, astronomical spectroscopy and remote sensing. There are two types of absorption: atomic absorption spectra and molecular absorption spectra.Ībsorption is used to determine the presence of a particular substance in a sample, or the quantity of the present substance in the sample. Absorption can be plotted in a wavelength, frequency or wave number. Absorption spectrum is the plotting of the energy that is absorbed by an element or substance. Absorption is used to determine the absorption level of certain objects and their ability to retain heat. Absorption is the ability of a matter or electron to absorb light or radiation which makes them transition into a higher energy state. The spectrum is a dark wavelength with bands of color on it, which is used to determine the emission of the object.Ībsorption is the opposite of emission, where energy, light or radiation is absorbed by the electrons of a particular matter. Emission can happen in the form of light and rays, such as gamma and radio. The frequencies can be determined using the formula Ephoton = hv, where ‘Ephoton’ is the energy of the photon, ‘v’ is its frequency, and ‘h’ is Planck's constant. The frequencies on a emission spectrum are recorded in light frequencies, where the color of the light determines the frequency. The amount of emission of an object varies depending on the spectroscopic composition of the object and temperature. The emittance of an object measures how much light is emitted by it. The emission of an element is recorded on an emission spectrum or atomic spectrum. The difference between the substance in ground state and excited state is then used to determine the emission level of the substance.Įach element or substances has a unique emission level or the amount of energy it radiates this helps the scientists identify elements in unknown substances. The molecules emit energy in the form of photons or light. 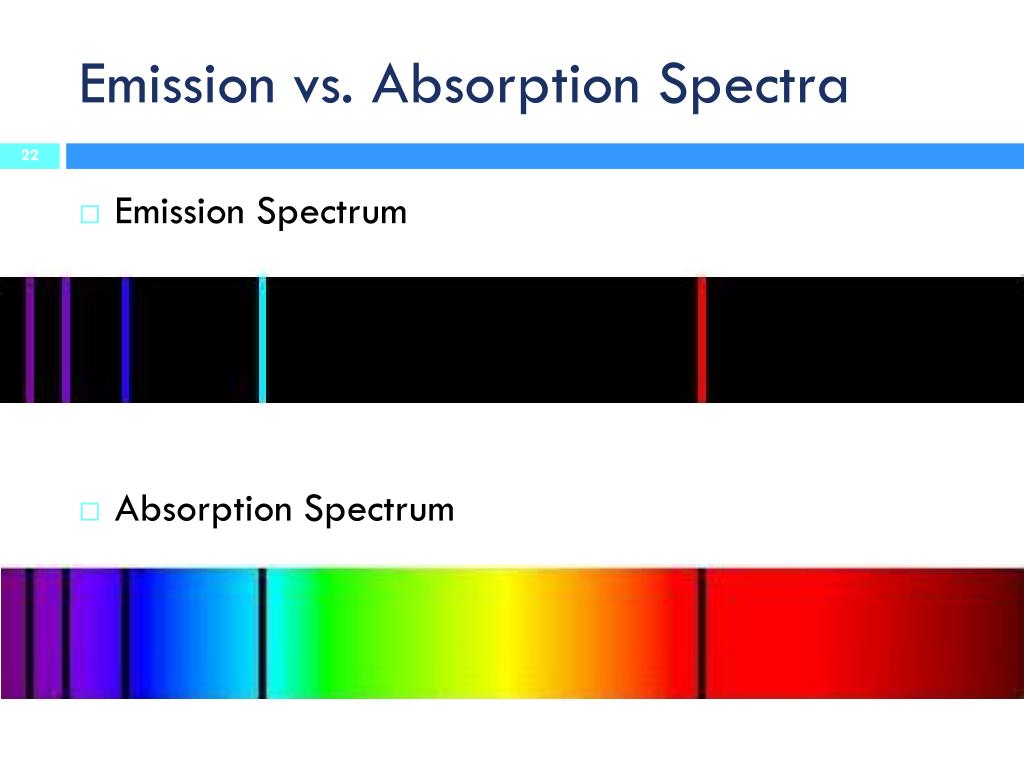
During this state the molecules are unstable and try to emit the energy in order to reach the state of equilibrium. 
However when heat, energy or light is applied to a substance, some of the molecules transition into a higher energy state or an excited state. The material starts off with being in the ground state, where all molecules are stable and settled. Every substances reacts differently when it interacts with light. Emission spectra and absorption spectra are different from each other but still related.Įmission is the ability of a substance to give off light, when it interacts with heat. This is really beneficial, of dealing with unknown substances. Using spectroscopy, a scientist can figure out the composition of a certain matter. Spectroscopy is the interaction of radiation and matter. Absorption is the opposite of emission, where energy, light or radiation is absorbed by the electrons of a particular matter.Įmission and absorption spectra are techniques that are used in chemistry and physics. Key Difference: Emission is the ability of a substance to give off light, when it interacts with heat.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
